Perplexing Pediatric Pancytopenia in Tanzania



Background
Pancytopenia has a broad differential in pediatrics,1,2 including infectious bone marrow suppression,3-5 bone marrow failure syndromes,6-8 and hematologic malignancies.9 While clinical features aid diagnostic evaluation, definitive diagnoses often require specialized testing that isn’t globally accessible.
Case Presentation
A 12-year-old male with no past medical history presented to a tertiary referral hospital in Dar es Salaam, Tanzania, with pancytopenia. Four months prior, he had intermittent high-grade fevers and generalized weakness, was diagnosed with malaria via rapid antigen test at a primary clinic (no species identified), and was treated with artemether/lumefantrine.
Two months later, he presented to a smaller tertiary hospital with abdominal distension and bilateral lower extremity. Abdominal ultrasound demonstrated organomegaly with portal vein dilatation (1.5 cm). Labs revealed leukopenia (white blood cell [WBC] 3.26 K/µl, absolute neutrophil count 1,370 cells/µl) and anemia (hemoglobin 7.4 g/dL) with normal platelets. Malaria, HIV, hepatitis B surface antigen, and stool analysis were negative; liver function tests and echocardiography were unavailable. Diagnosed with portal hypertension, he was started on spironolactone and oral iron before discharge.
One week later, he developed recurrent fever, epistaxis, hematemesis, and melena, requiring four units of packed red blood cells (pRBCs) at a district hospital. Upon transfer back to the initial tertiary hospital, he was pancytopenic (WBC <2 K/µl, hemoglobin 5.3 g/dL, platelet count 14 K/µl) and received three additional units of pRBCs, piperacillin/tazobactam, and tranexamic acid. Peripheral blood smear showed no abnormal morphology or hemolysis.
He was transferred to the larger tertiary referral hospital, where a bone marrow biopsy revealed normocellular marrow with preserved trilineage hematopoiesis (M:E 1:1) and no blasts or dysplasia. Iron stores and marrow fibrosis were not assessed. Flow cytometry was negative for leukemia. With supportive care and antibiotics, counts and ultrasound findings normalized within one week. (The sequence of events is summarized in the figure below).
Leading differentials included pancytopenia secondary to parasitic infection (Plasmodium falciparum) versus Gaucher disease (later ruled out) or medication-induced pancytopenia (less likely based on history and prescription records). However, in the absence of chromosomal breakage analysis, telomere length testing, or genetic panels, inherited bone marrow failure syndromes could not be excluded.
Discussion
This case illustrates how nonspecific symptoms and limited diagnostic testing creates uncertainty. Delayed marrow evaluation and genetic testing represent a global health equity issue. This case also highlights information transfer challenges across health system levels. Reliance on paper-based records and family-provided histories likely led to redundant testing and increased costs. Although hierarchical referrals aid triage, they may delay access to specialized care.
Once pancytopenia was identified, excluding hematologic malignancy was imperative given disease severity, treatment implications, and the risk of harm from empiric therapies such as glucocorticoids. However, a normocellular bone marrow biopsy should not halt evaluation, as alternative bone marrow failure etiologies early in disease progression, such as Fanconi anemia or GATA2 deficiency, were not yet ruled out.
The case further highlights that parasitic infections can cause bone marrow suppression — a finding more commonly attributed to viral illnesses. Parasites may rarely lead to pancytopenia through a hemophagocytic process that has been noted in a prior case report,10 whereas P. falciparum is more commonly associated with isolated anemia.5 In malaria-endemic regions, limited use of complete blood counts for uncomplicated malaria contributes to underrecognition of associated pancytopenia.
Finally, this case underscores the value of genetic testing, which was not available to evaluate for inherited causes of bone marrow failure. Conversely, access to available enzyme testing was critical in narrowing the differential diagnosis. Similarly, timely access to bone marrow biopsy at the tertiary referral hospital was crucial to excluding acute leukemia and guiding subsequent evaluation.
Figure. Timeline of case report
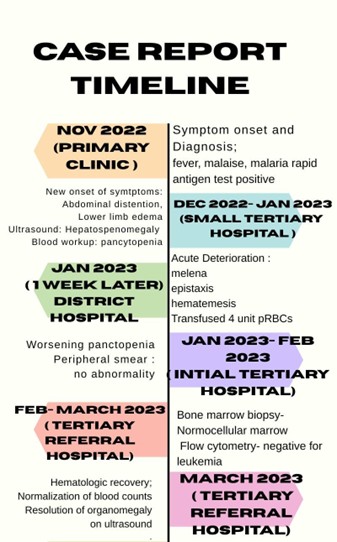
This timeline summarizes the health care referral hierarchy, clinical features and interventions of this case.
- Bhatnagar SK, Chandra J, Narayan S, et al. Pancytopenia in children: etiological profile. J Trop Pediatr. 2005;51(4):236-239.
- Naseem S, Varma N, Das R, et al. Pediatric patients with bicytopenia/pancytopenia: review of etiologies and clinico-hematological profile at a tertiary center. Indian J Pathol Microbiol. 2011;54(1):75-80.
- Young N, Mortimer P. Viruses and bone marrow failure. Blood. 1984;63(4):729-737.
- Pascutti MF, Erkelens MN, Nolte MA. Impact of viral infections on hematopoiesis: from beneficial to detrimental effects on bone marrow output. Front Immunol. 2016;7:364.
- Helleberg M, Goka BQ, Akanmori BD, et al. Bone marrow suppression and severe anaemia associated with persistent Plasmodium falciparum infection in African children with microscopically undetectable parasitaemia. Malar J. 2005;4:56.
- Alter BP. Diagnosis, genetics, and management of inherited bone marrow failure syndromes. Hematology Am Soc Hematol Educ Program. 2007;2007(1):29-39.
- Shimamura A, Alter BP. Pathophysiology and management of inherited bone marrow failure syndromes. Blood Rev. 2010;24(3):101-122.
- Nakao S. Diagnostic problems in acquired bone marrow failure syndromes. Int J Hematol. 2016;104(2):151-152.
- Gnanaraj J, Parnes A, Francis CW, et al. Approach to pancytopenia: diagnostic algorithm for clinical hematologists. Blood Rev. 2018;32(5):361-367.
- Zvulunov A, Tamary H, Gal N. Pancytopenia resulting from hemophagocytosis in malaria. Pediatr Infect Dis J. 2002;21(11):1086-1087.
Disclosure Statement: The doctors indicated no relevant conflicts of interest.
Acknowledgment: This article was edited by Ryan Mack, PhD, and Urshila Durani, MD, MPH.