Thrombosis and Vascular Biology
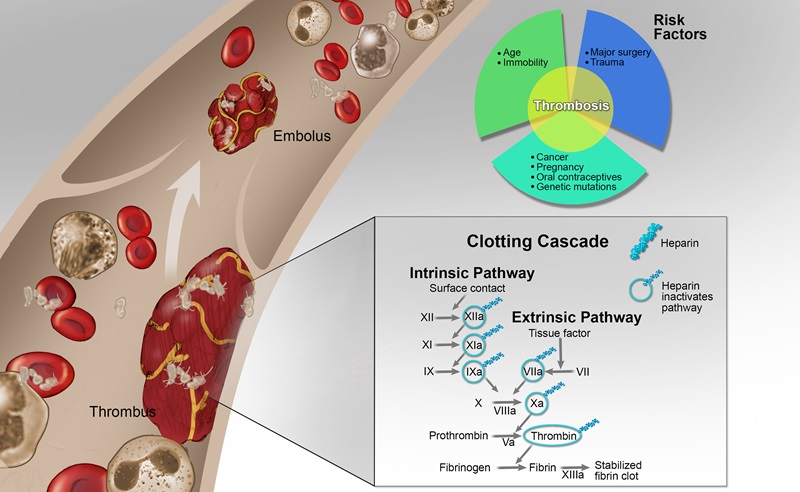
In spite of significant advances in understanding the interplay between vessels, blood, and hemodynamics in clotting, essential questions about thrombosis and thromboembolic disease persist. The emergence of COVID-19 infection and its association with thromboembolic risk underscores gaps in our understanding of the role of vasculopathy and endothelial dysfunction in hematologic disease.
- Enhance use of interdisciplinary technology to define the underlying pathophysiology of thrombosis, including the role of vascular dysfunction, blood composition and dysfunction, and hemodynamics in thrombogenesis and disease pathogenesis
- Develop new animal models of vasculopathy and thrombosis, and enhanced methods for imaging vasculopathy and thrombosis
- Define the contribution of coagulation end products to vascular development and function
- Improve clinical risk assessment, identification of biomarkers, development of scoring algorithms, and incorporation of pharmacogenomic data to assess the safety and efficacy of novel therapeutics