Development of Hematopoietic Progenitor and Stem Cells
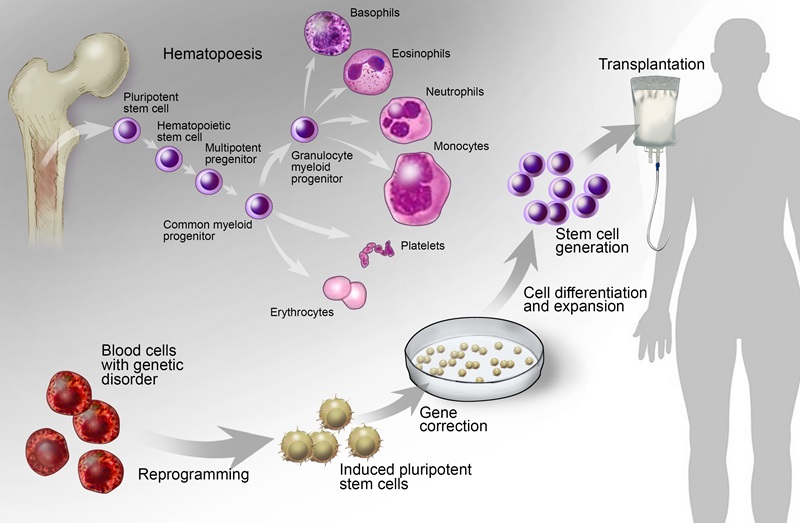
Our fundamental understanding of stem cell function in hematopoiesis is rapidly changing due to improved tools and new insights, including single cell “omics” technologies and high-resolution single cell and in situ imaging. Research in this area will fuel mechanistic discoveries and translational breakthroughs.
- Enhance fundamental understanding of stem cell self-renewal, regeneration and lineage commitment, and stem cell biology post-regeneration
- Understand how developmental hierarchy impacts normal and malignant hematopoiesis and how this can be used to target cancer cells with stem/progenitor features
- Delineate mechanisms of initiation, clonal expansion, and malignant progression in clonal hematopoiesis and how clonal hematopoiesis contributes to malignant and non-malignant diseases
- Test new approaches to hematopoietic stem cell transplantation, and new agents aimed to increased stem cell function, engraftment, and reconstitution